|
Diuretics are used to remove inappropriate water volume in animals with edema or volume overload, correct specific ion imbalances, and reduce blood pressure and pulmonary capillary wedge pressure (see Table: Dosages of Diuretics). They are classified by their mechanism of action as loop diuretics, carbonic anhydrase inhibitors, thiazides, osmotic diuretics, and potassium-sparing diuretics. The efficacy and use of each class of diuretic depends on the mechanism and site of action. Patterns of electrolyte excretion vary between classes, whereas maximal response is the same within a class. Therefore, if one drug within a class is ineffective, a different drug from the same class will likely be ineffective as well. Combining diuretics from different classes can lead to additive and potentially synergistic effects. See also Diuretics.
Topic Resources
Dosages of Diuretics
Furosemide:
Furosemide is a sulfonamide derivative and the most commonly administered diuretic in veterinary medicine. Furosemide is a loop diuretic; it inhibits the reabsorption of sodium and chloride in the thick, ascending loop of Henle, resulting in loss of sodium, chloride, and water into the urine. Furosemide induces beneficial hemodynamic effects before the onset of diuresis. Vasodilation increases renal blood flow, thereby increasing renal perfusion and lessening fluid retention. It appears that renal vasodilation depends on the local synthesis of prostaglandins.
The elimination half-life of furosemide is short in most animals (~15 min). The effect peaks 30 min after IV administration and 1–2 hr after PO administration. The duration of diuretic action is 2 and 6 hr after IV and PO administration, respectively. Furosemide is highly protein bound (91%–97%), almost totally to albumin. It is cleared through the kidneys by renal tubular secretion. Bioavailability of oral furosemide is low (only 50% is absorbed).

Furosemide is usually dosed to effect. For acute, short-term therapy, single IV, IM, or SC doses of 4–6 mg/kg are given. The major adverse effect from acute administration of large doses is acute intravascular volume reduction, which worsens cardiac output and hypotension and may precipitate acute renal failure. Chronic therapy in cats and some dogs can be accomplished by therapy every second or third day. Higher than normal doses of furosemide may be required in animals with renal disease due to functional abnormalities of the renal tubule and binding of furosemide to protein in the urine. If escalating doses of furosemide are required to control fluid retention, adding other types of volume-modifying medications, such as a potassium-sparing diuretic or an angiontensin-converting enzyme (ACE) inhibitor, may help avoid adverse effects.
Furosemide therapy is associated with a number of adverse effects. By nature of its mechanism of action, it causes dehydration, volume depletion, hypokalemia, and hyponatremia, which may be excessive and detrimental. Furosemide’s most important drug interaction is with the digitalis glycosides digoxin and digitoxin. The hypokalemia induced by furosemide diuresis potentiates digitalis toxicity. As long as animals continue to eat, hypokalemia does not usually develop. Hypokalemia also predisposes animals to hyponatremia by enhancing antidiuretic hormone secretion and the exchange of sodium ions for lost intracellular potassium ions. Concurrent administration of NSAIDs may interfere with furosemide's prostaglandin-controlled renal vasodilation and reduce the diuretic effect. Furosemide-induced dehydration of airway secretions may exacerbate respiratory disease.
Thiazide Diuretics:
The thiazide diuretics, hydrochlorothiazide and chlorothiazide, are not as potent as furosemide and thus are infrequently used in veterinary medicine. The thiazides act on the proximal portion of the distal convoluted tubule to inhibit sodium resorption and promote potassium excretion. They may be administered to animals that cannot tolerate a potent loop diuretic such as furosemide. They should not be administered to azotemic animals, because they decrease renal blood flow. Because the thiazides act on a different site of the renal tubule than other diuretics, they may be combined with a loop diuretic or potassium-sparing diuretic for treatment of refractory fluid retention. Adverse effects are electrolyte and fluid balance disturbances, similar to furosemide. Thiazides decrease renal excretion of calcium, so they should not be used in hypercalcemic animals.
Potassium-sparing Diuretics:
Potassium-sparing diuretics include spironolactone, amiloride, and triamterene. Spironolactone is the one most frequently used in veterinary medicine and is a competitive antagonist of aldosterone. Aldosterone is increased in animals with congestive heart failure when the renin-angiotensin system is activated in response to hyponatremia, hyperkalemia, and reductions in blood pressure or cardiac output. Aldosterone is responsible for increasing sodium and chloride reabsorption and potassium and calcium excretion from renal tubules. Spironolactone competes with aldosterone at its receptor site, causing a mild diuresis and potassium retention. Spironolactone is well absorbed after administration PO, especially if given with food. It is highly protein bound (>90%) and extensively metabolized by the liver to the active metabolite, canrenone. It is primarily eliminated by the kidneys. The onset of action for spironolactone is slow, and effects do not peak for 2–3 days. Spironolactone is not recommended as monotherapy but can be added to furosemide or thiazide therapy to treat cases of refractory heart failure. Because of the potential for hyperkalemia, spironolactone should not be administered concurrently with potassium supplements. It has been shown to be safe when used at low doses with concurrent ACE inhibitor therapy.
Sileo Dose In Cats FoodCarbonic Anhydrase Inhibitors:
Carbonic anhydrase inhibitors act in the proximal tubule to noncompetitively and reversibly inhibit carbonic anhydrase, which decreases the formation of carbonic acid from carbon dioxide and water. Reduced formation of carbonic acid results in fewer hydrogen ions within proximal tubule cells. Because hydrogen ions are normally exchanged with sodium ions from the tubule lumen, more sodium is available to combine with urinary bicarbonate. Diuresis occurs when water is excreted with sodium bicarbonate. As bicarbonate is eliminated, systemic acidosis results. Because intracellular potassium can substitute for hydrogen ions in the sodium resorption step, carbonic anhydrase inhibitors also enhance potassium excretion.
Osmotic Diuretics:
Osmotic diuretics include mannitol, dimethyl sulfoxide (DMSO), urea, glycerol, and isosorbide. Mannitol is commonly used in small animals but is expensive for use in adult large animals, so DMSO is often used in its place. Mannitol acts as a protectant against further renal tubular damage and initiates an osmotic diuresis. The initial dosage is 0.25–0.5 g/kg, given IV over 3–5 min. A response should be noted within 20–30 min. If a response is seen, the dose can be repeated every 6–8 hr, or a constant-rate infusion of 2–5 mL/min of a 5%–10% solution can be given. The total daily dosage should not exceed 2 g/kg. If diuresis is not seen, the initial dose can be repeated up to a total dosage of 1.5–2 g/kg. However, repeated doses usually are not more effective and increase the likelihood of complications (eg, edema).
DMSO is an oxygen-derived free radical scavenger and an osmotic diuretic. It is used in large animals to treat inflammatory and edematous conditions. It is a very potent solvent that can penetrate intact skin and carry other chemicals along with it. It penetrates all body tissues and produces an odor that many people cannot tolerate. The dosage is 1 g/kg, IV or via nasogastric tube, as a 10% solution diluted in 5% dextrose or lactated Ringer’s solution (higher concentrations can cause intravascular hemolysis).
It is a fact that herbs have been used by humans since time immemorial. Now, we have a long list of herbs with a myriad of uses and health benefits. This includes milk thistle, which is a thorny flowering plant that is native to the Mediterranean region. It is believed that the plant has been used to treat liver diseases in humans, for a very long time.
Apart from promoting the functions of the liver, this herb is also used for treating certain liver problems like cirrhosis, certain forms of hepatitis and mushroom poisoning. Studies show that the main component in milk thistle extract is silymarin, which is said to be beneficial for the liver. Apart from humans, animals too are said to benefit from this herb.
Similar to humans, liver is one of the vital organs for animals too. They are also prone to diseases of the liver, due to various reasons. Functioning of the liver can be affected with such diseases and in case of cats, the most common symptoms of liver disease include fatigue, loss of appetite, increased urination, bad breath, vomiting, pale-colored or gray-colored stools and weight loss.
In severe cases, the affected cats may develop swollen abdomen, confusion, seizures and jaundice. This condition can be fatal for the animals and so, early diagnosis and treatment is always beneficial. The common liver diseases in cats include feline hepatic lipidosis, toxic hepatopathy, cholangitis and portosystemic shunt. Milk thistle supplements can be used in such cats, as a part of supportive care.
Apart from protecting the liver from the damage that can be caused by free radicals, milk thistle is also said to be beneficial in strengthening the liver membranes. This herb is claimed to boost the rate of liver recovery and protect the organ from being bound by toxins. It is also believed that this herb stimulates growth of new liver cells, in place of damaged ones. This herb is recommended by vets for felines with liver conditions, like hepatitis, cholangitis, fatty liver, inflammatory liver disease, hepatomegaly and cancer.
As in case of other drugs, milk thistle dosage for cats may also vary from one feline to another. The right dosage is decided by the vet on the basis of various factors like the underlying cause, the condition of the cat, breed of the cat and its age and body size. This herb is now available in the form of pills/capsules and liquid extract. Make sure to purchase the product that is meant for cats and not those labeled as 'for veterinary use'. It has also been observed that liquid form of this herb is more preferred for cats, as it is easy to administer. It is also advisable to use milk thistle derivatives instead of silymarin-only supplements.
The dosage is decided on the basis of silymarin content. In case of cats, a single extract or powder capsule of 100 mg strength is used daily. This dose is for cats with a body weight of around 10 pounds. This dosage may be increased slightly for larger cats and those with severe liver problems.
On the other hand, the dosage will be reduced for cats with a body weight less than 10 pounds. In case of tablets with a strength of 250 mg, half a tablet will be sufficient. As far as liquid milk thistle for cats is concerned, the dosage may vary from 5 to 10 drops per day (divided into 2-3 times a day). In general, one drop of this extract is administered for every two pounds of the body weight of the cat.
Even though, there is no conclusive proof for the efficacy of this herb in felines, most of the pet owners are satisfied with its results. However, you must administer this herb to your pet cat, as per the instructions of the vet. He is the best person to determine whether the herb will prove effective for the feline and what will be the right milk thistle dosage for the animal. As mentioned above, the dosage may vary as per individual conditions. High doses of milk thistle may cause stomach upset and in such cases, lower the dose as per the vet's advice.
Disclaimer:This article is for informational purposes only and should not be used as a replacement for expert medical advice. So, follow the vet's instructions while administering herbal supplements like milk thistle for cats.
NMVMA listserve Veterinary Behavior Tip #19
Jeff Nichol, DVM Veterinary Behavior Medicine Veterinary Emergency and Specialty Centers
Albuquerque and Santa Fe Balloon phobia: avoidance, trazodone, & Sileo
There are dogs who are mildly nervous during the Balloon Fiesta and others who are so overwhelmed with panic that they scan the sky from the doorway. Similar to storm phobia cases, dogs who are wigged-out during this annual event are reactive to multiple sensory stimuli. Sound and sight can be equally frightening.
Consistent with any phobia, there is no logic in these dogs’ reactions. There is no desensitization/counterconditioning method that can make a difference in the immediate term. Here are the fundamentals of helping our clients with their completely freaked-out dogs over the next 10 days.
I hope this missive is helpful to you, your clients, and their pets.
All the best,
Jeff Nichol, DVM
This post may contain affiliate links.
What is SILEO?
I usually don't write about the press releases I receive; considering them spam and deleting with an eye roll. But this one caught my interest. The press release I received is an announcement of an upcoming gel called SILEO that was created by Zoetis, an animal heath company. This gel is supposed to help dogs that are afraid of loud noises: thunderstorms, constructions, and fireworks.
SILEO is a gel that is absorbed through the cheeks and gums (using a syringe) and after nearly an hour, your dog is feeling no pain. This is a way to dose your dog at home, helping him remain calm during a noise event, without making him drowsy and completely out of it.
When I read the press release, my first question is “what's in it?” I feed my dogs a raw food diet and you don't go through the time and study of creating and feeding a balanced, species-appropriate diet only to wreck it with chemicals.
The press release doesn't list the ingredients in the gel, but when you scroll down you'll find the Safety Information:
Do not use SILEO in dogs with severe cardiovascular disease, respiratory, liver or kidney diseases, or in conditions of shock, severe debilitation or stress due to extreme heat, cold or fatigue or in dogs hypersensitive to dexmedetomidine or to any of the excipients. SILEO should not be administered in the presence of preexisting hypotension, hypoxia or bradycardia. Do not use in dogs sedated from previous dosing. SILEO has not been evaluated in dogs younger than 16 weeks of age or in dogs with dental or gingival disease that could have an effect on the absorption of SILEO. SILEO has not been evaluated for use in breeding, pregnant or lactating dogs. Transient pale mucous membranes at the site of application may occur with SILEO use. Other uncommon adverse reactions included emesis, drowsiness or sedation. Handle gel-dosing syringes with caution to avoid direct exposure to skin, eyes or mouth.
I'm certain that the safety information isn't unusual. Now that drug companies can advertise on TV, we've come to expect that everything has the potential of either giving you horrible diarrhea when you're on the train or killing you. What struck me about the safety information is how different it looks from a natural supplement I use with my dogs that also alleviates fear and anxiety in dogs.
Click to follow me on Instagram at KeepTailWagging
What is Ewegurt?
Ewegurt is an all natural supplement that helps dogs relaxed when faced with fear, anxiety, and high-stress situations. I use Ewegurt when I know that there's going to be a thunderstorm, windstorm, power outage (power usually goes out during a windstorm), or before vet appointments.
If you visit Ewegurt.com, you can scroll down to see the ingredients:
The site calls it a treat, but I prefer to think of it as a supplement. I mix it into my dogs' food and 30-40 minutes later, they're doing good and the calm lasts for hours (with my dogs). I have been using this product for months and haven't noticed any side effects.
While many of us can predict fireworks, we can't predict thunderstorms; until now. We can shop for Weather Alert Radios at AcuRite.com! I watch the weather religiously and have the AccuWeather app on my phone. However, these aren't foolproof. The weather reports are typically for Seattle and we live 45 minutes north of Seattle. Getting an alert to storm trends is the perfect remind to add Ewegurt to our dogs' meals that morning and evening to keep them relaxed during thunderstorms.
 Is SILEO Safe for My Dogs?
Personally, I would prefer to use Ewegurt with my dogs, because it's all natural and it's a supplement that we've had success using with our dogs. However, this is a pretty convenient stance to have, because I'm not raising dogs that have severe anxiety. For dogs that have a dangerous level of anxiety, there may be a place for laboratory created products, when a natural solution (training, diet, and supplements) isn't the answer.
New edition music download. Listen to music from New Edition like Can You Stand The Rain, If It Isn't Love & more. Find the latest tracks, albums, and images from New Edition. Preview and download songs from New Edition by New Edition on iTunes. Songs from this album start at. Open iTunes to buy and download music.
So, do I think SILEO is safe for my dogs? No, I don't; but I have no proof to support my opinion. I'm not a veterinarian and I'm not a chemist. I'm a dog mom and dog nutrition blogger.
My distrust of this product stems from what I've learned about vaccinations, chemical flea & tick repellents,antibiotics, and other prescription medication that come with long-term side effects. I've become suspicious of anything that isn't natural because sometimes I feel that many in the pet industry seem to be more focused on taking our money than helping our dogs.
And no, I'm not wearing a tin foil hat. I'm just terrified of the unnecessary use of chemicals on our dogs and the long-term impact on the health of our fur kids.
Sadly, there isn't a perfect answer for every dog and I'm thankful that I have been able to avoid harsh chemicals with my dogs. If you think SILEO may be something that will help your dog, I encourage you to go over the risks with your veterinarian so that you can make steps to protect your dog.
UPDATE on SILEO Ingredients: Visit my post on keeping our dogs safe on the Fourth for more information about SILEO, including safety tests completed by the brand.
This page contains information on Sileo for veterinary use.The information provided typically includes the following:
SileoThis treatment applies to the following species:Company: Zoetis
NADA 141-456, Approved by FDA
(dexmedetomidine oromucosal gel)
Each mL of SILEO contains 0.09 mg dexmedetomidine (equivalent to 0.1 mg dexmedetomidine hydrochloride).
For oromucosal use in dogs only. Not intended for ingestion.
Sileo Caution
Federal law (USA) restricts this drug to use by or on the order of a licensed veterinarian.
Description
SILEO (dexmedetomidine oromucosal gel) is a synthetic alpha-2 adrenoceptor agonist. Each ml of SILEO contains 0.09 mg dexmedetomidine (equivalent to 0.1 mg dexmedetomidine hydrochloride). The chemical name is (+)-4-[1-(2,3-dimethylphenyl) ethyl]-1H-imidazole monohydrochloride. It is a white, or almost white, crystalline, water soluble substance having a molecular weight of 236.7. The molecular formula is C13 H16 N2 HCl and the structural formula is:
Sileo Indications
SILEO is indicated for the treatment of noise aversion in dogs.
Dosage and Administration
Always provide the Client Information Sheet with SILEO and instruct the client how to properly operate the SILEO dosing syringe (see Client Information Sheet for Owner/Handler Use and Safety).
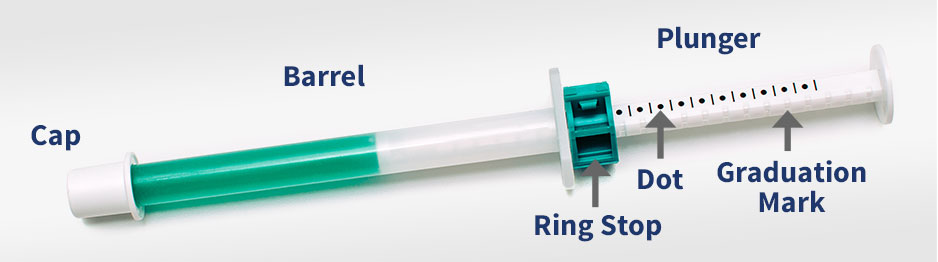
To prevent accidental overdose, it is important to make sure that the ring-stop on the dosing syringe is in the locked position prior to administration.
SILEO is administered onto the oral mucosa between the dog’s cheek and gum at the dose of 125 mcg/m2. The gel is absorbed through the oral mucosa and therefore it should NOT be swallowed. If the gel is swallowed, the product may not be effective. If the gel is swallowed, do not repeat the dose for at least two hours.
The following table provides the acceptable dosage for each weight range. Each dot (•) represents 0.25 mL of SILEO.
Each ml contains the equivalent to 0.1mg dexmedetomidine hydrochloride.
Table 1. Dosage by body weight
If the dose is more than 6 dots, divide the dose between both sides of the mouth.
The first dose of SILEO should be administered approximately 30-60 minutes before the fear and/or anxiety-eliciting noise stimulus, immediately after the dog shows first signs of anxiety or fear related to noise, or when the owner detects a typical noise stimulus (e.g. sound of fireworks) eliciting anxiety or fear in the dog. Typical signs of anxiety and fear associated with noise aversion are panting, trembling, pacing, seeking people, trying to hide, trying to escape, freezing behavior, refusing to eat food or treats, inappropriate urination or defecation, and salivation.
Administering SILEO without use of the SILEO syringe will result in incorrect dosing, which may result in lack of efficacy or overdose.
Dosing should be performed by an adult. Impermeable disposable gloves should be worn when administering SILEO and when handling the SILEO syringe.
If noise lasts longer than 2-3 hours and the dog’s signs of fear and/or anxiety reappear, another dose may need to be given. To avoid overdosing, there should always be at least two hours’ pause between dosages. No more than 5 doses can be given during one noise event.
A partially used syringe can be used again within 4 weeks after initial opening, if there is enough gel for a complete dose for the dog. To minimize the risk of incorrect dosing, a partially used syringe that does not have enough gel for a complete dose should not be used.
Any unused product or waste material should be disposed of according to local law and Federal prescription drug disposal guidelines.
Contraindications
Do not use SILEO in dogs with severe cardiovascular, respiratory, liver or kidney disease, or in conditions of shock, severe debilitation, or stress due to extreme heat, cold or fatigue. Do not use in dogs with hypersensitivity to dexmedetomidine or to any of the excipients.
Warnings
Human Safety: Not for human use. Keep out of reach of children.
Avoid administering the product if pregnant, as exposure may induce uterine contractions and/or decrease fetal blood pressure.
Appropriate precautions should be taken while handling and using filled syringes. Impermeable disposable gloves should be worn when handling the syringe, administering SILEO, or when coming in contact with the dog’s mouth after application.
If skin is damaged, dexmedetomidine can be absorbed into the body. In case of skin contact, wash with soap and water. Remove contaminated clothing.
SILEO can be absorbed following direct exposure to skin, eyes, or mouth. In case of accidental eye exposure, flush with water for 15 minutes. If wearing contact lenses, eyes should be rinsed first, then remove contact lenses and continue rinsing, then seek medical advice immediately.
Accidental exposure may cause sedation and changes in blood pressure. In case of accidental exposure, seek medical attention immediately. Exposure to the product may induce a local or systemic allergic reaction in sensitized individuals.
Note to physician: This product contains an alpha-2 adrenoceptor agonist.
The safety data sheet (SDS) contains more detailed occupational safety information. To report adverse reactions in users or to obtain a copy of the SDS for this product call 1-888-963-8471.
Animal Safety: SILEO should not be administered in the presence of pre-existing hypotension, hypoxia, or bradycardia. Sensitive dogs may experience a drop in body temperature and heart rate, and may appear sedated. These dogs should be kept warm and not offered food or water until SILEO’s effects have worn off (usually within a few hours). Do not use in dogs sedated from previous dosing.
Precautions
SILEO is not meant to be swallowed. Instead, it must be placed onto the mucosa between the dog’s cheek and gum. If SILEO is swallowed, the product may not be effective. If SILEO is swallowed, do not repeat the dose for at least two hours. Feeding and giving treats within 15 minutes after administration should be avoided.
The use of other central nervous system depressants may potentiate the effects of SILEO.
As with all alpha-2 adrenoceptor agonists, the potential for isolated cases of hypersensitivity, including paradoxical response (excitation), exists.
SILEO has not been evaluated in dogs younger than 16 weeks of age or in dogs with dental or gingival diseases that could have an effect on SILEO’s absorption. SILEO has not been evaluated for aversion behaviors to thunderstorms.
The safety and effectiveness of SILEO in breeding, pregnant, and lactating dogs has not been evaluated. Administration to pregnant dogs may induce uterine contractions and/or decrease fetal blood pressure.
Adverse Reactions
In a well-controlled European field study, which included a total of 182 dogs ranging from 2 to 17 years of age and representing both mixed and pure breed dogs (89 treated with dexmedetomidine oromucosal gel and 93 treated with control), no serious adverse reactions were attributed to administration of dexmedetomidine oromucosal gel.
Table 2 shows the number of dogs displaying adverse reactions (some dogs experienced more than one adverse reaction).
Table 2. Adverse Reactions - Number (%) of dogs
Pale mucous membranes were frequently seen in dogs treated with dexmedetomidine oromucosal gel. In most cases, the effect was transient and no adverse reactions due to mucosal irritation were reported.
In a second well-controlled European field study which included a total of 36 dogs ranging from 2 to 17 years of age and representing both mixed and pure breed dogs (12 treated with dexmedetomidine oromucosal gel at 125 mcg/m2, 12 treated with dexmedetomidine oromucosal gel at 250 mcg/m2, and 12 treated with a vehicle control), no serious adverse reactions were attributed to administration of dexmedetomidine oromucosal gel.
Table 3 shows the number of dogs displaying adverse reactions (some dogs experienced more than one adverse reaction).
Table 3. Adverse Reactions - Number (%) of dogs
To report suspected adverse events, for technical assistance or to obtain a copy of the SDS call 1-888-963-8471.
For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1-888-FDA-VETS or online at http://www.fda.gov/AnimalVeterinary/SafetyHealth
Clinical Pharmacology
Dexmedetomidine is a potent and selective alpha-2 adrenoceptor agonist that inhibits the release of noradrenaline from noradrenergic neurons. A reduction of heart rate may occur. In a clinical safety study the mean heart rate dropped from 127 bpm to 98 bpm for dogs treated with the dexmedetomidine gel.
Sileo Dose In Cats Treatment
Oral bioavailability of dexmedetomidine is poor due to extensive first-pass metabolism. No measurable concentrations were found after gastro-intestinal tubing of dexmedetomidine to dogs. When administered via the oral mucosa, enhanced bioavailability is observed as a result of absorption in the oral cavity and the avoidance of first-pass metabolism in the liver.
The maximum concentration of dexmedetomidine occurs at about 0.6 hours after both IM and oromucosal administration in dogs. In a pharmacokinetic study in dogs, the oromucosal mean bioavailability of dexmedetomidine was 28%. The apparent volume of distribution of the drug is 0.9 L/kg. In the canine circulation, dexmedetomidine is largely bound to plasma proteins (93%).
Dexmedetomidine is eliminated by biotransformation mainly in the liver with a half-life ranging from 0.5 to 0.7 hours after intravenous (IV) administration and 0.5 to 3 hours after oromucosal administration in dogs. Metabolism accounts for more than 98% of the elimination. Known metabolites show no or negligible activity. Metabolites are excreted mainly in the urine with a minor fraction found in the feces.
ANIMAL SAFETY:
Canine safety study with dexmedetomidine hydrochloride injectable
In a laboratory study of dexmedetomidine hydrochloride injection, doses of 375, 1125 and 1875 mcg/m2 were administered once daily IV or doses of 500, 1500 and 2500 mcg/m2 [low, mid, and high doses, respectively] were administered once daily IM on three consecutive days.
Dexmedetomidine-induced sedation occurred on each dosing day at all dose levels from 2 to 8 hours post-dosing. The duration of sedation increased with the dosage level. A decreased respiratory rate was observed in all groups on each day, but returned to normal within 4 hours in the low IV and IM groups and within 8 hours in the mid and high IV and IM groups. A decreased rectal temperature was observed in all groups on each day. In the low IV and IM groups, the rectal temperature returned to normal or near normal within 4 hours and in the mid and high IV and IM groups within 8 hours. A decreased heart rate was observed in all groups on each day, with greater decreases in the IV dosing groups. In the low groups, the duration of decrease was 2 hours with the values returning to normal 4 hours after dosing. In the mid groups, the duration of decrease was 4 hours with the values returning to normal 8 hours after dosing. In the high IV dose group, the values also returned to normal at 8 hours after dosing. However, in the high IM group, the values were still slightly depressed at 8 hours after dosing; the values returned to normal by 24 hours after dosing. On electrocardiography, QT time was prolonged in all groups due to decreased heart rate. In the low and mid IM groups, one case each of second degree A-V-block was observed. In the high IM group, one dog had first and second degree A-V block.
In the mid IV group, two dogs had second degree A-V block and one dog had first and second degree A-V block. In the mid and high IM groups, one dog each had elevated alanine aminotransferase (ALT) values. In the high IV group, two dogs had elevated ALT values.
Then try to run a game and get a new error message. Pcx2 file not found isofilesystem. 'PS2 BIOS ERROR'.
Dexmedetomidine was well tolerated in the study even at the high doses, and adverse effects on physiology were related to the pharmacology of the drug. There were no toxicological effects on body weight, clinical variables, gross or microscopic pathology.
Effectiveness
SILEO was evaluated in a European randomized multi-center, double-blind, vehicle-controlled field study. Effectiveness was evaluated in client-owned dogs, ranging in age between 2 and 17 years, and in body weight between 4 and 67 kg. A total of 187 dogs were randomized, 144 of which were evaluated for effectiveness: 71 dogs received 125 mcg/m2 of dexmedetomidine oromucosal and 73 dogs received the vehicle control gel up to 5 times as needed with a minimum interval of 2 hours between doses. All dogs had in previous years suffered from acute fear and/or anxiety due to fireworks.
The first dose was given on New Year’s Eve before or as soon as the dog showed the first signs of becoming anxious and/or fearful due to fireworks. Re-dosing could be performed as soon as the dog again demonstrated noise aversion behaviors, but at least 2 hours after the previous dose to allow time to assess observable effects of treatment and to avoid potential cumulative effects of dexmedetomidine.
For the first co-primary endpoint, the proportion of dogs with owner-assessed good or excellent treatment effect was higher in dogs treated with dexmedetomidine oromucosal gel (53/71 dogs) than in those administered control (24/73 dogs). The proportion of dogs with some, no, or worse effect was higher in dogs administered control (49/73 dogs) than in those treated with dexmedetomidine (18/71 dogs). Refer to Table 4. below.
Table 4. Owner assessment of treatment effect/score by treatment group
There was a statistically significant difference (p<0.0001) between dexmedetomidine and control in favor of dexmedetomidine. The odds ratio was 5.5876, with 95% confidence interval (2.7635, 11.2976).
For the second co-primary variable, the mean sum of behavior scores over the treatment period was significantly different between dexmedetomidine and control (p=0.0069). Behavior scores consisted of a cumulative score using a number of outward signs of noise aversion in dogs such as panting, trembling, vocalizing, and seeking people. The mean score was lower (better) for the dexmedetomidine oromucosal gel treated group than for the control group (LSMeans: 4.9661, 7.2456 respectively). Refer to Table 5 below.
Table 5. Mean behavioral sum scores by treatment and time point
Of the different types of behaviors, dogs treated with dexmedetomidine oromucosal gel displayed less panting, trembling and trying to hide than those treated with the control gel.
How Supplied
SILEO is packaged in HDPE dosing syringe enabling doses from 0.25 to 3 ml. The syringe is fitted with plunger, dosing ring and end cap. Each syringe is further packed into a carton with a label and a leaflet.
Package sizes: (1 syringe per carton) 1 x 3 ml, 3 x 3 ml, 5 x 3 ml, 10 x 3 ml, 20 x 3 ml.
Not all package sizes may be marketed.
STORAGE INFORMATION:
Store unopened and opened syringes in the original package at controlled room temperature 20-25°C (68-77°F) with excursions permitted to 15-30°C (59-86°F). Use syringe contents within 4 weeks after opening the syringe.
SILEO® is a trademark of Orion Corporation.
Mfd by: Orion Corporation, Turku, Finland
Dist by: Zoetis Inc., Kalamazoo, MI 49007
Made in Finland
Revised: November 2017
148211-5
CPN: 3690520.2
ZOETIS INC.333 PORTAGE STREET, KALAMAZOO, MI, 49007
Copyright © 2019 Animalytix LLC. Updated: 2019-06-28
Comments are closed.
|
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
- Blog
- Blog
- New Page
- Home
- Onyx Productionhouse X 10.0.0.89.iso
- Portal Test Chamber 19
- Download Amelinha 1998 Amelinha
- Programming 25q64fvsig Bios Spi Hp
- Modern Firearms Gunsmith Edition
- Samp Unde Se Tuneaza Tahoma
- Dynamics Nav Free Download
- Gta San Andreas Tow Truck Mod
- Naruto Ultimate Ninja Storm 4 Multiplayer
- Blackweb Keyboard Color Change
- White Day Game Download
- Ip Man 1 Full Movie In Hindi Free Download Filmywap
- Blackshot Games Download Free
- J Ring Glass Studio
- New Lord Of The Rings Game 2019
- Max Payne 3 Chapter 7
- Russo Japanese War Propaganda
- Cannot Remove Apple Software Update Windows 10
- Call Voice Changer Intcall Apk Hack
 RSS Feed
RSS Feed
